what happens to the molecules of a compound when it changes from liquid to solid
Chapter 10. Solids and Liquids
Phase Transitions: Melting, Humid, and Subliming
- Describe what happens during a phase alter.
- Calculate the energy change needed for a phase change.
Substances tin can change stage — often because of a temperature modify. At low temperatures, well-nigh substances are solid; every bit the temperature increases, they become liquid; at higher temperatures still, they become gaseous.
The process of a solid becoming a liquid is chosen melting (an older term that you may see sometimes is fusion). The contrary process, a liquid becoming a solid, is called solidification. For any pure substance, the temperature at which melting occurs — known equally the melting point — is a characteristic of that substance. It requires energy for a solid to melt into a liquid. Every pure substance has a certain amount of energy it needs to change from a solid to a liquid. This corporeality is chosen the enthalpy of fusion (or rut of fusion) of the substance, represented as ΔH fus. Some ΔH fus values are listed in Table x.two "Enthalpies of Fusion for Various Substances"; it is assumed that these values are for the melting point of the substance. Note that the unit of measurement of ΔH fus is kilojoules per mole, then we demand to know the quantity of cloth to know how much energy is involved. The ΔH fus is always tabulated equally a positive number. However, it tin can be used for both the melting and the solidification processes as long equally you lot go on in mind that melting is ever endothermic (so ΔH will exist positive), while solidification is e'er exothermic (and then ΔH will be negative).
| Substance (Melting Point) | ΔH fus (kJ/mol) |
|---|---|
| Water (0°C) | 6.01 |
| Aluminum (660°C) | 10.vii |
| Benzene (5.5°C) | 9.95 |
| Ethanol (−114.three°C) | 5.02 |
| Mercury (−38.8°C) | two.29 |
What is the energy modify when 45.7 g of HiiO melt at 0°C?
Solution
The ΔH fus of H2O is 6.01 kJ/mol. However, our quantity is given in units of grams, non moles, and then the first step is to catechumen grams to moles using the molar mass of H2O, which is xviii.0 thou/mol. Then we can use ΔH fus as a conversion cistron. Because the substance is melting, the process is endothermic, so the energy change volition have a positive sign.
![]()
Without a sign, the number is assumed to be positive.
Examination Yourself
What is the energy change when 108 g of C6H6 freeze at 5.5°C?
Answer
−13.viii kJ
During melting, energy goes exclusively to changing the phase of a substance; it does not get into changing the temperature of a substance. Hence melting is an isothermal process because a substance stays at the same temperature. But when all of a substance is melted does any boosted energy get to irresolute its temperature.
What happens when a solid becomes a liquid? In a solid, private particles are stuck in place because the intermolecular forces cannot exist overcome by the energy of the particles. When more energy is supplied (due east.g., by raising the temperature), in that location comes a point at which the particles have enough energy to motion around only not plenty energy to separate. This is the liquid phase: particles are still in contact merely are able to move around each other. This explains why liquids can assume the shape of their containers: the particles move effectually and, under the influence of gravity, make full the lowest volume possible (unless the liquid is in a nada-gravity environment — see Figure 10.16 "Liquids and Gravity").

The phase change between a liquid and a gas has some similarities to the phase change betwixt a solid and a liquid. At a certain temperature, the particles in a liquid accept plenty energy to get a gas. The procedure of a liquid becoming a gas is chosen boiling (or vapourization), while the process of a gas becoming a liquid is called condensation. Notwithstanding, unlike the solid/liquid conversion procedure, the liquid/gas conversion process is noticeably afflicted past the surrounding pressure on the liquid because gases are strongly affected by pressure. This ways that the temperature at which a liquid becomes a gas, the humid point, tin alter with surrounding force per unit area. Therefore, we define the normal boiling point every bit the temperature at which a liquid changes to a gas when the surrounding pressure is exactly 1 atm, or 760 torr. Unless otherwise specified, information technology is assumed that a humid point is for 1 atm of force per unit area.
Like the solid/liquid phase change, the liquid/gas stage change involves energy. The amount of energy required to catechumen a liquid to a gas is called the enthalpy of vaporization (or oestrus of vaporization), represented every bit ΔH vap. Some ΔH vap values are listed in Table 10.3 "Enthalpies of Vaporization for Various Substances"; it is causeless that these values are for the normal boiling bespeak temperature of the substance, which is also given in the table. The unit for ΔH vap is besides kilojoules per mole, and so we need to know the quantity of textile to know how much energy is involved. The ΔH vap is also e'er tabulated as a positive number. Information technology can be used for both the boiling and the condensation processes as long as you keep in mind that boiling is always endothermic (and so ΔH volition be positive), while condensation is always exothermic (so ΔH volition be negative).
| Substance (Normal Boiling Point) | ΔH vap (kJ/mol) |
|---|---|
| H2o (100°C) | twoscore.68 |
| Bromine (59.5°C) | 15.4 |
| Benzene (fourscore.ane°C) | 30.8 |
| Ethanol (78.3°C) | 38.six |
| Mercury (357°C) | 59.23 |
What is the energy alter when 66.7 g of Brii(g) condense to a liquid at 59.5°C?
Solution
The ΔH vap of Brtwo is 15.4 kJ/mol. Even though this is a condensation procedure, we can notwithstanding use the numerical value of ΔH vap equally long as nosotros realize that we must accept energy out, so the ΔH value will be negative. To determine the magnitude of the energy change, nosotros must first convert the amount of Br2 to moles. Then we can apply ΔH vap as a conversion factor.
![]()
Because the process is exothermic, the actual value will be negative: ΔH = −6.43 kJ.
Exam Yourself
What is the energy change when 822 g of C2H5OH(ℓ) boil at its normal boiling betoken of 78.3°C?
Answer
689 kJ
As with melting, the energy in boiling goes exclusively to changing the stage of a substance; information technology does not go into changing the temperature of a substance. And then humid is also an isothermal procedure. Merely when all of a substance has boiled does any additional energy go to changing its temperature.
What happens when a liquid becomes a gas? We accept already established that a liquid is equanimous of particles in contact with each other. When a liquid becomes a gas, the particles separate from each other, with each particle going its own way in space. This is how gases tend to fill their containers. Indeed, in the gas stage virtually of the book is empty infinite; only about i ane-thousandth of the volume is actually taken up by matter (see Effigy 10.17 "Liquids and Gases"). It is this property of gases that explains why they can be compressed, a fact that is considered in Chapter six "Gases".
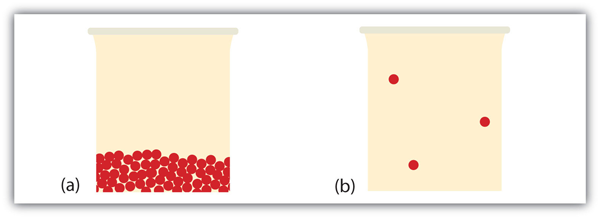
Nether some circumstances, the solid phase tin can transition directly to the gas phase without going through a liquid phase, and a gas tin directly become a solid. The solid-to-gas change is called sublimation, while the contrary process is called deposition. Sublimation is isothermal, like the other stage changes. There is a measurable energy change during sublimation; this energy change is called the enthalpy of sublimation, represented as ΔH sub. The human relationship between the ΔH sub and the other enthalpy changes is as follows:
ΔH sub = ΔH fus + ΔH vap
As such, ΔH sub is not always tabulated considering it can exist merely calculated from ΔH fus and ΔH vap.
There are several mutual examples of sublimation. A well-known product — dry water ice — is actually solid CO2. Dry ice is dry because it sublimes, with the solid bypassing the liquid phase and going direct to the gas phase. The sublimation occurs at temperature of −77°C, so it must be handled with caution. If you have ever noticed that ice cubes in a freezer tend to go smaller over time, information technology is because the solid water is very slowly subliming. "Freezer burn" isn't actually a burn; it occurs when certain foods, such as meats, slowly lose solid h2o content because of sublimation. The nutrient is all the same good merely looks unappetizing. Reducing the temperature of a freezer will slow the sublimation of solid h2o.
Chemical equations can exist used to represent a stage change. In such cases, it is crucial to utilize phase labels on the substances. For example, the chemical equation for the melting of ice to brand liquid water is as follows:
H2O(s) → HtwoO(ℓ)
No chemical change is taking identify; however, a physical change is taking place.
Heating Curves
A plot of the temperature versus the amount of heat added is known every bit a heating bend (encounter Effigy 10.18). These are ordinarily used to visually prove the human relationship between phase changes and enthalpy for a given substance.
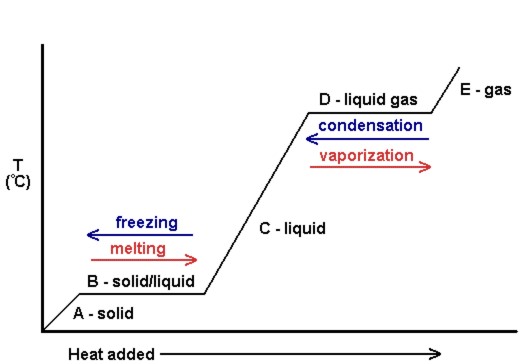
In Figure ten.xviii[i], the solid gains kinetic free energy and consequently rises in temperature as heat is added. At the melting bespeak, the heat added is used to break the attractive intermolecular forces of the solid instead of increasing kinetic free energy, and therefore the temperature remains constant. Later on all the solid has melted, again, the heat added goes to increasing the kinetic energy (and temperature) of the liquid molecules until the boiling indicate. At the humid point, one time again, the heat added is used to intermission the bonny intermolecular forces instead of supplying kinetic free energy, and the temperature remains abiding until all liquid has been turned to gas.
- Phase changes can occur between whatever two phases of matter.
- All phase changes occur with a simultaneous change in energy.
- All phase changes are isothermal.
Questions
- What is the difference between melting and solidification?
- What is the difference between boiling and condensation?
- Depict the molecular changes when a solid becomes a liquid.
- Describe the molecular changes when a liquid becomes a gas.
- What is the free energy alter when 78.0 m of Hg melt at −38.8°C?
- What is the energy change when 30.8 grand of Al solidify at 660°C?
- What is the energy modify when 111 g of Br2 boil at 59.v°C?
- What is the energy change when 98.6 grand of H2O condense at 100°C?
- Each of the following statements is incorrect. Rewrite them so they are correct.
- Temperature changes during a phase change.
- The process of a liquid becoming a gas is called sublimation.
- Each of the post-obit statements is wrong. Rewrite them so they are correct.
- The book of a gas contains only about ten% matter, with the rest being empty infinite.
- ΔH sub is equal to ΔH vap.
- Write the chemical equation for the melting of elemental sodium.
- Write the chemical equation for the solidification of benzene (C6H6).
- Write the chemical equation for the sublimation of COtwo.
- Write the chemical equation for the humid of propanol (CthreeH7OH).
- What is the ΔH sub of H2O? (Hint: see Tabular array 10.ii "Enthalpies of Fusion for Various Substances" and Table 10.iii "Enthalpies of Vaporization for Various Substances".)
- The ΔH sub of Iii is 60.46 kJ/mol, while its ΔH vap is 41.71 kJ/mol. What is the ΔH fus of Itwo?
Answers
- Melting is the phase alter from a solid to a liquid, whereas solidification is the phase change from a liquid to a solid.
- The molecules have enough energy to move most each other merely not plenty to completely separate from each other.
- 890 J
- 10.7 kJ
-
- Temperature does not change during a phase alter.
- The process of a liquid becoming a gas is called humid; the procedure of a solid becoming a gas is chosen sublimation.
- Na(s) → Na(ℓ)
- CO2(s) → COii(g)
- 46.69 kJ/mol
Media Attributions
- "Glass of Water" © 2005 by Derek Jensen is licensed under a Public Domain license
- "Clayton Anderson zero g" © 2010 by NASA is licensed under a Public Domain license
Source: https://opentextbc.ca/introductorychemistry/chapter/phase-transitions-melting-boiling-and-subliming/
0 Response to "what happens to the molecules of a compound when it changes from liquid to solid"
Post a Comment